|
6/16/2023 0 Comments Atom meaning/atom-57e1bb583df78c9cce33a106.jpg)
If an atom has more or fewer electrons than protons, then it has an overall negative or positive charge, respectively-such atoms are called ions. If the number of protons and electrons are equal, then the atom is electrically neutral. The protons have a positive electric charge, the electrons have a negative electric charge, and the neutrons have no electric charge. More than 99.94% of an atom's mass is in the nucleus. Atoms are so small that accurately predicting their behavior using classical physics is not possible due to quantum effects. This is smaller than the shortest wavelength of visible light, which means humans cannot see atoms with conventional microscopes. A human hair is about a million carbon atoms wide. The number of neutrons defines the isotope of the element.Ītoms are extremely small, typically around 100 picometers across. For example, any atom that contains 11 protons is sodium, and any atom that contains 29 protons is copper. The atom is the basic particle of the chemical elements, and the chemical elements are distinguished from each other by the number of protons that are in their atoms. Smallest recognized division of a chemical elementĮlectrons and a compact nucleus of protons and neutronsĪn atom is a particle that consists of a nucleus of protons and neutrons surrounded by a cloud of electrons. 
The black bar is one angstrom ( 10 −10 m or 100 pm). 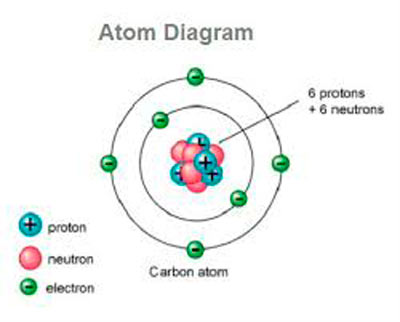
The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. An illustration of the helium atom, depicting the nucleus (pink) and the electron cloud distribution (black).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
